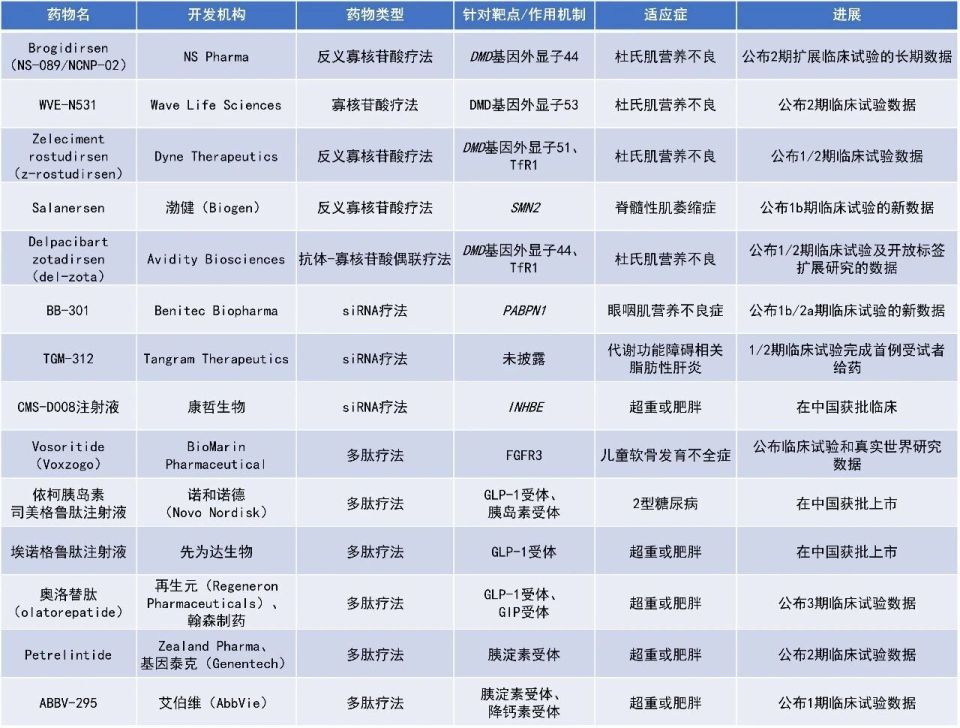
近期,全球多肽和寡核苷酸(TIDES)领域迎来系列进展。诺和诺德(Novo Nordisk)基础胰岛素/胰高糖素样肽-1受体激动剂(GLP-1RA)周制剂依柯胰岛素司美格鲁肽注射液、先为达生物cAMP偏向型GLP-1RA埃诺格鲁肽注射液在中国获批上市。再生元(Regeneron Pharmaceuticals)公布了其与翰森制药合作开发的GLP-1/葡萄糖依赖性促胰岛素多肽(GIP)双受体激动剂奥洛替肽(olatorepatide)的3期临床试验的积极顶线数据。本文将节选其中部分重要进展做简单介绍,仅供读者参阅。
依柯胰岛素司美格鲁肽注射液:在中国获批上市

近日,中国国家药品监督管理局(NMPA)正式批准了依柯胰岛素司美格鲁肽注射液(商品名:诺和杰)在中国的上市申请。新闻稿指出,诺和杰是全球首个获批的基础胰岛素/GLP-1RA周制剂,由胰岛素周制剂依柯胰岛素和GLP-1RA周制剂司美格鲁肽组成,适用于接受基础胰岛素或GLP-1RA治疗后血糖控制不佳的成人2型糖尿病患者,在饮食和运动基础上联合口服降糖药物进行治疗。
诺和杰的两个组分,基础胰岛素和GLP-1RA联合治疗能够发挥协同互补优势,实现在增强疗效的同时减少胰岛素治疗带来的体重增加和低血糖风险。研究结果显示,在已使用基础胰岛素治疗的成人2型糖尿病患者中,诺和杰组糖化血红蛋白(HbA1c)<7%的达标率是基础胰岛素组的2倍(72% vs 36%);低血糖发生率相比基础胰岛素组降低78%,体重获益明确。此外,诺和杰的司美格鲁肽组分具有多效性作用,在血糖控制之外,可能协助改善成人2型糖尿病患者的其他代谢危险因素。COMBINE 1和COMBINE 3研究的附加分析显示,接受诺和杰治疗的患者在腰围、血压和血脂谱方面均观察到较基线改善,且相较于胰岛素治疗,其改善的幅度更大。
埃诺格鲁肽注射液:在中国获批上市

先为达生物宣布,cAMP偏向型GLP-1受体激动剂埃诺格鲁肽注射液获得NMPA批准,适用于在控制饮食和增加体力活动基础上对成人超重/肥胖患者的长期体重管理。基于cAMP偏向型GLP-1受体激动机制,埃诺格鲁肽注射液可高选择性激活cAMP信号通路、最大程度减少β-arrestin募集,与传统非偏向型GLP-1RA形成明显差异。这一机制直接带来两大核心优势:一是减重效果强效持久,全程无明显平台期;二是在降低体重的同时,同步改善代谢、减少慢病风险。
埃诺格鲁肽注射液减重适应症的获批主要基于其在中国超重或肥胖成人中开展的一项3期临床试验SLIMMER。基于疗法策略,第40周时,埃诺格鲁肽呈现出剂量依赖性体重降低,最高剂量(2.4 mg)至第48周时,体重降幅提升至15.4%(安慰剂校正后为15.1%),92.8%的受试者实现≥5%的临床意义体重下降。79.6%和63.5%的受试者分别达到了≥10%和≥15%的体重下降。接受埃诺格鲁肽治疗的患者在48周治疗结束时,体重仍在持续下降,未达到平台期,这表明延长埃诺格鲁肽治疗时间可能实现更大的体重降幅。除显著体重降低外,埃诺格鲁肽组可显著改善其他关键的心血管代谢风险指标,包括腰围、血压、血脂、HbA1c、空腹血糖、胰岛素水平和胰岛素敏感性标志物HOMA-IR。平均尿酸水平降幅高达54.3 µmol/L,高尿酸血症发生率较安慰剂组更低。此外,埃诺格鲁肽可显著降低肝脏脂肪含量,在基线肝脏脂肪含量≥8%的受试者中,埃诺格鲁肽2.4 mg组第40周时肝脏脂肪含量较基线的平均百分比变化达到-53.1%。所有剂量治疗组的肝酶水平均较安慰剂组显著降低。
奥洛替肽:公布3期临床试验数据

再生元宣布,其合作伙伴翰森制药公布了奥洛替肽在中国开展的3期临床试验的积极顶线数据。该研究评估了奥洛替肽用于治疗成人肥胖或超重患者的疗效与安全性。奥洛替肽是一种新型GLP-1/GIP双受体激动剂。根据双方的战略授权协议,再生元拥有在中国大陆、香港和澳门以外地区的独家临床开发和商业权益,并计划于2026年晚些时候启动全球3期注册性临床项目。
这项随机、双盲、安慰剂对照3期试验共纳入604名成人患者,评估了每周一次皮下注射5 mg、10 mg或15 mg奥洛替肽与安慰剂在48周内的效果。研究达到共同主要终点:与安慰剂相比,奥洛替肽显著降低体重,并使更高比例的患者实现至少5%的体重减轻。数据显示,接受奥洛替肽治疗的患者在第48周平均体重减轻达19%,97%的患者体重下降≥5%。此外,奥洛替肽展现出良好的胃肠道耐受性,恶心和呕吐的平均发生率分别低于10%和5%。
一体化平台助力多肽药物开发
多肽药物已经成为重要创新药物模式,优化其药物代谢与药代动力学(DMPK)特征,已成为推动候选分子成功进入临床阶段的关键环节之一。与传统小分子相比,多肽往往面临酶降解快、体内暴露不足、清除率高及跨膜转运受限等挑战,因此需要以机制为导向的系统化DMPK策略。围绕这些特点,药明康德DMPK团队构建了面向多肽分子的综合优化体系,通过整合体外稳定性研究、体内药代评价与生物分析技术,支持研发团队在早期阶段即识别影响药代行为的关键因素,并实现数据驱动的分子优化决策。
基于多肽DMPK策略专题研究与实践经验,团队通过选择与生理环境高度相关的研究体系,对多肽在全血、血浆、溶酶体及肝肾S9等多种生物基质中的稳定性与代谢路径进行系统评估,从机制层面解析降解模式与清除机制,为序列修饰与递送策略优化提供依据。同时,结合跨物种体内PK研究与组织分布分析,可进一步建立体外与体内结果之间的关联关系,提升药代预测的可靠性,并支持剂量设计与临床转化评估。
在实验实施层面,药明康德通过优化样品处理流程与分析方法,解决多肽易吸附、稳定性不足及低暴露检测难度高等常见技术挑战,从而确保关键数据的准确性与可重复性。依托标准化研究流程与跨学科协作模式,该体系能够在发现、候选确定及IND前研究阶段持续提供决策支持,帮助研发团队在分子设计、制剂策略与给药方案之间实现协同优化。通过将机制理解与系统化实验平台相结合,药明康德DMPK团队有效提升多肽药物DMPK特征的可预测性与可开发性,为下一代多肽创新疗法的高效推进奠定坚实基础。
参考资料:
[1] Benitec Biopharma Announces Positive Interim Phase 1b/2a Results for High Dose BB-301 and Continued Durable Improvements for Low Dose BB-301 Treatment at the 2026 Muscular Dystrophy Association Clinical & Scientific Conference. Retrieved March 13, 2026, from https://www.globenewswire.com/news-release/2026/03/09/3251690/0/en/Benitec-Biopharma-Announces-Positive-Interim-Phase-1b-2a-Results-for-High-Dose-BB-301-and-Continued-Durable-Improvements-for-Low-Dose-BB-301-Treatment-at-the-2026-Muscular-Dystroph.html
[2] AbbVie Announces Positive Topline Results from a Phase 1 Multiple Ascending Dose Study of ABBV-295, a Long-Acting Amylin Analog, in Adults. Retrieved March 13, 2026, from https://www.prnewswire.com/news-releases/abbvie-announces-positive-topline-results-from-a-phase-1-multiple-ascending-dose-study-of-abbv-295-a-long-acting-amylin-analog-in-adults-302707602.html
[3] Dyne Therapeutics Announces New Positive Cardiopulmonary Results from DELIVER Trial of Z-Rostudirsen in Duchenne Muscular Dystrophy (DMD). Retrieved March 9, 2026, from https://www.globenewswire.com/news-release/2026/03/08/3251476/0/en/Dyne-Therapeutics-Announces-New-Positive-Cardiopulmonary-Results-from-DELIVER-Trial-of-Z-Rostudirsen-in-Duchenne-Muscular-Dystrophy-DMD.html
[4] Biogen Presents Additional Salanersen Data Showing New Motor Milestones Achieved in Children with SMA Previously Treated with Gene Therapy. Retrieved March 11, 2026, from https://investors.biogen.com/news-releases/news-release-details/biogen-presents-additional-salanersen-data-showing-new-motor
[5] 全球首个偏向型GLP-1减重药物先维盈®获批上市! Retrieved March 6, 2026, from https://mp.weixin.qq.com/s/GNGCsMIOiyiqoOxDLeN5tQ
[6] Genentech Announces Positive Phase II Results for Petrelintide, an Amylin Analog Developed for People Living With Overweight and Obesity. Retrieved March 5, 2026, from https://www.businesswire.com/news/home/20260305198579/en/Genentech-Announces-Positive-Phase-II-Results-for-Petrelintide-an-Amylin-Analog-Developed-for-People-Living-With-Overweight-and-Obesity
[7] 康哲药业自主研发创新药INHBE小核酸药物CMS-D008 获得超重或肥胖适应症药物临床试验批准. Retrieved March 4, 2026, from https://mp.weixin.qq.com/s/fJ-Xf0K-WS7b2KyBdywsPw
[8] Olatorepatide Obesity Treatment Licensed by Regeneron Demonstrates Positive Phase 3 Results in Chinese Patients. Retrieved March 9, 2026, from https://www.globenewswire.com/news-release/2026/03/09/3251688/0/en/Olatorepatide-Obesity-Treatment-Licensed-by-Regeneron-Demonstrates-Positive-Phase-3-Results-in-Chinese-Patients.html
[9] 全球首个基础胰岛素/GLP-1RA周制剂诺和杰®在中国获批,中国将成为全球首个商业上市国家. Retrieved March 6, 2026, from https://www.prnasia.com/story/524345-1.shtml
[10] Brogidirsen (NS-089/NCNP-02) 4.5-Year Clinical Trial Data for the Treatment of Duchenne Muscular Dystrophy Presented at 2026 MDA Clinical & Scientific Conference. Retrieved March 9, 2026, from https://www.prnewswire.com/news-releases/brogidirsen-ns-089ncnp-02-4-5-year-clinical-trial-data-for-the-treatment-of-duchenne-muscular-dystrophy-presented-at-2026-mda-clinical--scientific-conference-302708276.html
[11] Del-zota treatment is associated with near normalization of CK levels and improvements in key functional outcomes at 1 year in participants with DMD44. Retrieved March 12, 2026, from https://www.mdaconference.org/abstract-library/del-zota-treatment-is-associated-with-near-normalization-of-ck-levels-and-improvements-in-key-functional-outcomes-at-1-year-in-participants-with-dmd44/
[12] Wave Life Sciences Reports Fourth Quarter and Full Year 2025 Financial Results and Provides Business Update. Retrieved March 12, 2026, from https://ir.wavelifesciences.com/news-releases/news-release-details/wave-life-sciences-reports-fourth-quarter-and-full-year-2025
[13] Tangram Therapeutics Announces First Participant Dosed in Phase 1/2 RESTORE-MASH Trial of TGM-312, a Novel Investigational RNAi Medicine. Retrieved March 5, 2026, from https://www.globenewswire.com/news-release/2026/03/05/3250112/0/en/Tangram-Therapeutics-Announces-First-Participant-Dosed-in-Phase-1-2-RESTORE-MASH-Trial-of-TGM-312-a-Novel-Investigational-RNAi-Medicine.html
[14] BioMarin Presents New Data on the Positive Impact of Early Treatment With VOXZOGO® (vosoritide) on Proportionality and Arm Span in Children with Achondroplasia at the 2026 American College of Medical Genetics and Genomics (ACMG) Annual Clinical Genetics Meeting. Retrieved March 12股票配资平台门户, 2026, from https://www.prnewswire.com/news-releases/biomarin-presents-new-data-on-the-positive-impact-of-early-treatment-with-voxzogo-vosoritide-on-proportionality-and-arm-span-in-children-with-achondroplasia-at-the-2026-american-college-of-medical-genetics-and-genomics-acmg-a-302711657.html
牛策略提示:文章来自网络,不代表本站观点。